Description of Procedure or Service
This policy only addresses coverage of testosterone in adult individuals (age 18 years and older)
Testosterone is a naturally occurring lipophilic androgen hormone that is produced by both males and females for various functions. In males, testosterone is produced by the interstitial cells of Leydig in the testis. In females, testosterone is primarily created and disseminated by the ovaries and adrenal glands. Testosterone is required for synthesis of dihydrotestosterone (DHT) as well as estradiol (E2). Sex hormone-binding globulin (SHBG) binds testosterone to aid in transport and intratesticular bioavailability.
Dysregulation in testosterone levels can lead to serious conditions, including hypogonadism and other testosterone excess or deficiency conditions. Additional hormones, including follicle-stimulating hormone (FSH), luteinizing hormone (LH), and prolactin, play roles in development. As part of the hypothalamic-pituitary-gonadal axis, FSH and LH bind to gonadal receptors to modulate testosterone. During conditions of dyshomeostasis, such as hypogonadism, FSH, LH, and prolactin serum levels can be used as diagnostic tools.
Terms such as male and female are used when necessary to refer to sex assigned at birth.
***Note: This Medical Policy is complex and technical. For questions concerning the technical language and/or specific clinical indications for its use, please consult your physician.
Policy
BCBSNC will provide coverage for testosterone when it is determined the medical criteria or reimbursement guidelines below are met
Benefits Application
This medical policy relates only to the services or supplies described herein. Please refer to the Member's Benefit Booklet for availability of benefits. Member's benefits may vary according to benefit design; therefore, member benefit language should be reviewed before applying the terms of this medical policy.
When testosterone is covered
- Reimbursement for measurement of serum total testosterone (see Note 1) is allowed for any of the following:
- For symptoms of androgen deficiency or androgen excess in males:
- For initial screening, two measurements at least 24 hours apart.
- If the initial screening was normal but symptoms persist, follow-up testing is allowed no sooner than 60 days after the initial screening.
- For the monitoring of treatment response in men taking enzyme inhibitors for prostate cancer.
- For men receiving testosterone replacement therapy (every 2-3 months for the first year after initiation of therapy or after a change in therapeutic dosage; annually thereafter).
- For gender-dysphoric/gender-incongruent persons (baseline, during treatment, and for therapy monitoring).
- For symptomatic females (see Note 2) being evaluated for conditions associated with androgen excess (e.g., polycystic ovary syndrome and functional hypothalamic amenorrhea).
- For symptoms of androgen deficiency or androgen excess in males:
- Reimbursement for annual measurement of serum free testosterone, sex hormone-binding globulin (SHBG), and/or albumin is allowed for males with total testosterone confirmed as low or borderline low and who have hypogonadism, gynecomastia, and/or other forms of testicular hypofunction.
- Reimbursement for individuals suspected of having a disorder that is accompanied by increased or decreased SHBG levels (see Notes 3 and 4), measurement of serum free testosterone using a medically accepted algorithm based on total serum testosterone, SBHG, and/or albumin or bioavailable testosterone is allowed.
- Reimbursement is allowed prior to initiating testosterone therapy for males with gynecomastia, for once per lifetime serum estradiol testing.
- Reimbursement is allowed for individuals with ambiguous genitalia, hypospadias, or microphallus, measurement of serum dihydrotestosterone for the diagnosis of 5-alpha reductase deficiency.
When testosterone is not covered
- Reimbursement is not allowed in asymptomatic individuals or in individuals with non-specific symptoms for measurement of serum total testosterone, free testosterone, and/or bioavailable testosterone.
- Reimbursement is not allowed for measurement of serum free testosterone and/or bioavailable testosterone as primary testing (i.e., in the absence of prior serum total testosterone measurement).
- Reimbursement is not allowed for the use of saliva for the measurement of testosterone.
- Reimbursement is not allowed for the identification of androgen deficiency in women, measurement of serum testosterone.
- Reimbursement for all other situations not mentioned above, measurement of serum dihydrotestosterone is not allowed.
Note 1: Serum total testosterone sample collection should occur in the early morning, after fasting. Due to considerable variability in serum total testosterone levels, the Centers for Disease Control and Prevention (CDC) developed a standardization program for total testosterone assays (Hormone Standardization [HoSt]/Testosterone). An assay certified by the CDC’s HoSt/Testosterone program is standardized to within ±6.4% of the CDC total testosterone reference standard. It is STRONGLY RECOMMENDED that serum total testosterone measurement be performed with an assay that has been certified by the CDC HoSt/Testosterone program. A list of CDC-certified assays is available on the HoSt website.
Note 2: When measuring serum total testosterone in females, please note that the technology used for measurement must be sensitive enough to detect the low serum total testosterone levels that are normally found in females.
Note 3: Conditions associated with decreased SHBG concentrations according to the 2018 Endocrine Society Guidelines:
- Obesity
- Diabetes mellitus
- Use of glucocorticoids, progestins, and androgenic steroids
- Nephrotic syndrome
- Hypothyroidism
- Acromegaly
- Polymorphisms in the SHBG gene
Note 4: Conditions associated with increased SHBG concentrations according to the 2018 Endocrine Society Guidelines
- Aging
- HIV disease
- Cirrhosis and hepatitis
- Hyperthyroidism
- Use of some anticonvulsants
- Use of estrogens
- Polymorphisms in the SHBG gene
Policy Guidelines
The steroid hormone, testosterone, plays a role in both male and female development and health. In males, testosterone is involved in the stage-specific differentiation of germ cells, spermatogenesis, and the synthesis of dihydrotestosterone (DHT) and estradiol (E2). DHT stimulates sexual differentiation of male genitalia during embryogenesis, genital maturation during puberty, and growth of pubic and facial hair. E2 is required in males for modulating libido, erectile function, and spermatogenesis. Serum testosterone is typically solubilized by binding to the androgen binding protein (ABP) or SHBG, which aids in regulating their transport, distribution, metabolism, and biological activity. ABP and SHBG have similar primary structure, but they differ in the types of oligosaccharides associated with them.
In females, testosterone is primarily synthesized and secreted in the ovaries and adrenal glands but some testosterone production also occurs in peripheral tissues like muscle, fat, breast, and bone. Polycystic ovary syndrome (PCOS) is one manifestation of a dysregulation of testosterone in women and is a complicated condition with a variety of metabolic, reproductive, and psychological features.
Primary and secondary hypogonadism are two forms of testicular hypofunction found in males. These conditions can be differentiated by the concentration of serum LH, FSH, and prolactin. Primary hypogonadism is associated with low levels of testosterone and normal to high levels of LH and FSH. Secondary hypogonadism is associated with low levels of testosterone and normal to low levels of LH and FSH. The anterior pituitary gland of hypothalamic-pituitary-gonadal axis releases LH and FSH, which act on the gonadal receptors to regulate testosterone production. Binding of LH to Leydig cell receptors initiates testosterone production, while testosterone secretion is further regulated by feedback inhibition. Males who develop hypogonadism prior to puberty often exhibit depressed secondary sex characteristics, eunuchoid stature, small testes, gynecomastia, and a small phallus. For males who develop hypogonadism after the onset of puberty, the physical findings are similar, except for a normal stature and normal phallus size. Besides hypogonadism, testosterone production can also be affected by certain medications, chemotherapy, lifestyle, and aging.
In adult males, total serum testosterone levels decrease at an average rate of 1.6% per year. The concentrations of free and bioavailable testosterone decrease more rapidly, typically two-three percent annually, due to the natural increase in SHBG. By the age of 60, 20% of men will have testosterone levels below the normal range, “and the figure rises to 50% in those aged over 80.” Significant decrease in testosterone may result in symptoms such as fatigue, decreased libido, erectile dysfunction, depression, muscle weakness, and others. Unfortunately, these symptoms are not specific to testosterone deficiency. Low testosterone levels are associated with diabetes, metabolic syndrome, cardiovascular disease, obesity, sleep apnea, and other disorders. Additionally, testosterone elevations are associated with serious conditions including tumors, hyperthyroidism, and genetic disorders such as congenital adrenal hyperplasia.
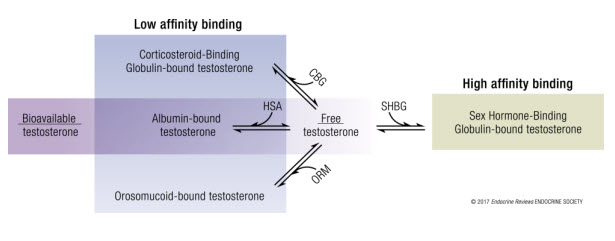
Within the serum, testosterone can be either free (i.e. not bound to a specific protein) or protein-bound. Only one to four percent of circulating testosterone is usually found free. SHBG binds testosterone with a high affinity whereas serum albumin, corticosteroid-binding globulin (CBG), and orosomucoid binds testosterone with a much lower affinity. “Bioavailable” testosterone refers to the amount of free testosterone and albumin-bound testosterone as indicated in the figure below.
CDC Hormone Standardization (HoSt) Program-Testosterone
Serum testosterone testing can measure either total testosterone (TT) concentration, free testosterone, or bioavailable testosterone. TT can be “measured using radioimmunoassay, immunometric assays, or liquid chromatography–tandem mass spectrometry. Considerable inter-assay and inter-laboratory variability is often found in TT measurements. When 1133 laboratories using 14 different assays measured TT concentrations using the same College of American Pathologists quality control sample from a single hypogonadal man, the measured values ranged from 45 to 365 ng/dL (1.6 to 12.7 nmol/L).”
The Centers for Disease Control and Prevention (CDC) released their analysis of TT in serum by isotope dilution-liquid chromatography-tandem mass spectrometry (ID-LC-MS/MS) in 2012. As a part of the CDC HoSt Program (CDC Hormone Standardization Program) to certify and calibrate hormone assays, the CDC monitors and validates hormone testing by laboratories and manufacturers. “Calibration is further verified by analyzing serum material with assigned reference values for total testosterone every six months and comparing the results obtained against predefined acceptance limit, which is ± 6.4 % from the target value.” According to CDC standards, TT using ID/HPLC/MS/MS methodology has a reportable range of 2.5 – 1000 ng/dL or 0.09 – 34.7 nM with a limit of detection of 0.36 ng/dL or 0.012 nM. As for accuracy in terms of “trueness and precision,” the CDC reports that total serum testosterone precision (%CV) ranges from 2.2% to 5.5%. The following limitation was noted: “This method was tested for total testosterone analysis in human serum and may not be suitable for other specimens such as plasma, whole blood, urine, and/or saliva. The analytical performance parameters need to be reassessed and verified when other specimen matrices are used.” A list of the assays certified by the CDC HoSt/Testosterone program can be found on the CDC’s HoSt website.
Analytical Validity
A recent study used a liquid chromatography with tandem mass spectrometry (LC-MS-MS) method to assess salivary testosterone, androstenedione, dehydroepiandrosterone, and 17-OH-progesterone. The authors state that the accuracy of this method is “between 83.0 and 106.1% for all analytes” and conclude that this “LC-MS/MS method allowed a sensitive evaluation of androgen salivary levels and represents an optimal technique to explore the relevance of a comprehensive androgen profile as measured in saliva for the study of androgen secretion modulation and activity in physiologic and pathologic states.” However, another study compared various ELISA-based salivary testosterone assays and noted that “proportional errors between the methods calls [sic] for caution” as one of the four methods yielded no results due to malfunction. Another study compared salivary testosterone measurements using immunoassays with those measured by tandem mass spectrometry. The authors conclude that the immunoassay-based methods “tended to inflate estimates of lower testosterone concentrations.”
Recently, van der Veen, et al. (2019) developed and validated a LC-MS/MS method to establish reliable reference intervals in five plasma steroid hormones (progesterone, 17-hydroxyprogesterone, androstenedione, testosterone and dihydrotestosterone); these researchers utilized samples from 280 healthy male and female participants over a four-month period. Women taking oral contraceptive pills were found to have lower levels of 17-OH-progesterone and androstenedione; further, it was identified that hormonal biological variation was typically greater in women compared to men. Final conclusions stated that “The gender-specific determination of the reference intervals, together with the observation that the biological variation demonstrated a high degree of variation, allows interpretation of data on individual and group level for improved biochemical characterization of patients in clinical practice."
Star-Weinstock and Dey (2019) developed an accurate and sensitive method to measure testosterone in hypogonadal adults and children of both genders; this quantification method utilized electrospray ionization (ESI)-LC-MS/MS and achieved a “sensitivity of one ng/dL from 100 µL sample volume.” The authors note that two highlights of this novel method are that this sample preparation technique “includes simultaneous protein precipitation and derivatization,” and that this TT measurement method was certified by the CDC Hormone Standardization program.
Sun, et al. (2020) developed and validated an isotope dilution ultra-performance liquid chromatography-tandem mass spectrometry method (ID-UPLC-MS/MS) to measure human serum testosterone. This method offers higher accuracy and lower variability than the traditional immunoassays, especially when measuring low testosterone levels in hypogonadism. To assess accuracy of the method, pure testosterone was added to the serum samples and the actual concentrations after two serial liquid-liquid extractions were measured. The actual concentrations were close to the female and male levels, with a recovery rate ranging from 94.32 to 108.6%. Sensitivity, specificity, and precision were also measured and met the performance criteria standards established by Clinical and Laboratory Standards Institute and the Hormone Standardization Program of the Center for Disease Control. “Moreover, the [ID-UPLC-MS/MS] method exhibited a good consistency between low and high concentrations of testosterone. In addition, the method required a simple sample preparation and a small sample volume, therefore it may be suitable for routine clinical practice."
The Centers for Disease Control and Prevention (CDC) reviewed testosterone testing in 2014 in a group of 6746 participants of various age groups and both sexes. The positive bias identified by steroid analyte testing indicated that the test was measuring additional compounds (and not only the analyte in question). The authors concluded that “although technologies for steroid hormone measurement have advanced significantly, measurement variability within and across laboratories has not improved accordingly… Within-assay variability for current assays is generally high, especially at low analyte concentrations."
Testosterone and other hormones (AMH, FSH, LH, free androgen index (FAI), prolactin, estradiol) have been used for the clinical diagnosis of polycystic ovary syndrome (PCOS). In a study completed by Khashchenko, et al. (2020), 130 girls with PCOS had the accuracy and specificity of hormonal testing assessed and cutoffs for the most significant hormone indicators of PCOS diagnosis in adolescents were identified. The authors found that “Levels of testosterone > 1.15 nmol/L, androstenedione > 11.45 ng/mL, and LH/FSH ratio > 1.23 also showed high sensitivity of 63.2–78.2% and specificity of 84.4–93.7% in PCOS diagnosis in the studied sample of girls.” The combined use of either four thresholds (AMH, FAI, testosterone, androstenedione, LH/FSH ratio as previously stated) yielded a diagnostic accuracy of 90.2-91.6% in predicting PCOS in adolescents.
Dalmiglio, et al. (2024) evaluated the Vermeulen formula as less expensive and more accessible method of assessing FT than equilibrium dialysis or ultrafiltration. The study included 190 patients, all of whom received FT measurements through both direct immunoluminometric assay and the Vermeulen formula. The authors claim that “the calculated method employing the Vermeulen formula was considered the gold standard.” The authors noted that the sensitivity was lower in females, which they claim could be because of a potential proportional bias and the low number of true positive cases. The authors concluded that “the direct method exhibited comparable performance to the calculated method, but caution should be exercised when interpreting results, particularly in females."
Clinical Utility and Validity
Equilibrium dialysis is the gold standard for determining free serum testosterone. Unfortunately, it is technically difficult and has limited availability. Compared to other less accurate methods, it is expensive. It relies on the accuracy and precision of TT determination. In equilibrium dialysis, a semipermeable membrane is used to retain the bound testosterone on one side of the membrane while the free testosterone equilibrates between the two sides. It is dependent on environmental conditions including pH, ionic strength, and temperature; in fact, steroids, such as testosterone, can bind up to 2.5 times higher at 4◦C than at 37◦C. One study shows that increasing the temperature from 37◦C to 41◦C increased the free cortisol level by approximately 80%.
Immunoassays to measure free and bioavailable testosterone are inaccurate. The Endocrine Society urges the use of medically accepted algorithms that rely on TT, SHBG, and/or albumin to estimate serum free testosterone. Multiple algorithms have been published. The recent allosteric model proposed by Zakharov and colleagues models the binding of testosterone as a multi-step, dimeric process. This allosteric model has “close correspondence with those measured using equilibrium dialysis."
A 2017 international study comprised of multiple cohorts with healthy, non-obese males attempted to “derive standardized, age-specific reference ranges” for circulating testosterone; it was stated that “a substantial proportion of intercohort variation in testosterone levels is due to assay differences.” Further, the issue in developing standards for circulating testosterone due to variation in body mass and comorbidities was also noted. “Another unresolved issue relates to whether the reference sample should include only the healthy nonobese men or whether it should include the entire population of men 19 to 39 years. Obesity and comorbid conditions affect circulating TT concentrations; therefore, inclusion of obese men with comorbid conditions could distort the reference ranges. Whether the reference ranges generated in nonobese men are appropriate for use in obese men deserves further investigation. Even though men with known diagnoses of conditions or diseases associated with hypogonadism were excluded, it is possible a small percentage of individuals in these cohorts may be hypogonadal.”
Shukla, et al. (2018) organized a cross-sectional study to measure the relationship between prostatic specific antigen (PSA) and serum testosterone levels in both healthy men and men with partial androgen deficiency (PADAM); a total of 255 men participated in this study. “Mean total testosterone and serum PSA was 9.35 ± 1.33 nmol/L, 1.96 ± 0.76 ng/mL in males with PADAM and 15.30 ± 1.95 nmol/L, 1.85 ± 0.73 ng/mL respectively in males without PADAM. No significant relationship was observed between serum PSA and serum testosterone levels among healthy males irrespective of PADAM.” Results from this study suggest that PSA values do not need to be adjusted “for biopsy decisions according to testosterone levels."
In a retrospective cohort study, eighty-five severely hypogonadal men were observed for changes in serum PSA concentrations during testosterone treatment for 18 months. The Endocrine Society clinical guidelines recommend measuring PSA in hypogonadal men over the age of 50 at three months and twelve months after starting testosterone therapy and urologic referral if serum PSA > 1.4 ng/mL above baseline or to an absolute value > four ng/mL. Studies have been performed in men with mild to moderate hypogonadism which reported smaller increases in serum PSA concentrations during testosterone treatment; however, no studies have reported serum PSA changes in response to testosterone treatment of severely hypogonadal men. In this study, testosterone treatment “increased the median serum testosterone concentration from 36 ng/dL at baseline to 395 ng/dL at 6-18 months. This treatment resulted in a median increment in PSA above baseline of 0.70 ng/mL at 6-18 months…31% of men had increases in PSA >1.4 ng/mL; and 13% of men reached absolute PSA concentrations > four ng/mL." The authors suggest that “testosterone treatment of severely hypogonadal men often increases PSA above the commonly accepted thresholds for urologic referral [and] that future clinical guidelines for the expected PSA response to testosterone replacement reflect the degree of hypogonadism."
A total of nine years of registry data, comprised of 650 patients with hypogonadism, was analyzed to determine the impact of long-term intramuscular testosterone treatment (1000 mg every 10-12 weeks). Serum testosterone concentrations were found to increase “from 5.7±2.3 nmol/L to 19.4±2.8 nmol/L in men with classical hypogonadism and from 7.8±2.4 nmol/L to 19.2±3.1 nmol/L in men with functional hypogonadism”; final conclusions suggest that patients with the functional form of hypogonadism may benefit the most from testosterone treatment as “men with functional hypogonadism were more likely to lose ten percent weight and five percent of waist circumference (WC) than men with classical hypogonadism." Men with functional hypogonadism were also more likely to be obese at the start of the study.
Cauley, et al. (2021) performed a study to examine the effect of testosterone treatment on TBS. “Two hundred and eleven men were enrolled in Bone Trial of the Testosterone Trials. Of these, 197 men had two repeat TBS and vBMD measurements; 105 men were allocated to receive testosterone, and 92 men to placebo for one year. TBS, a BMD, and vBMD were assessed at baseline and month 12.” The results of this study report that there was no difference in the percent change in TBS by randomized group. They saw a 1.6% (95% confidence intervals (CI) 0.2–3.9) change in the testosterone group and a 1.4% (95% CI−0.2, 3.1) change in the placebo group. In contrast, they saw a six percent increase in vBMD (95% CI4.5–7.5) in the testosterone group as compared to only a 0.4% vBMD change (95% CI−1.65–0.88) in the placebo groups. As a result, the authors concluded that TBS was not clinically useful in monitoring the one year effect of testosterone treatment on the bone structure in older hypogonadal men.
Stern and Casto (2024) studied the differences in salivary testosterone levels across the menstrual cycle. A total of 339 people with a menstrual cycle and confirmed ovulation were included. Salivary testosterone was measured with LC-MS/MS four times across the mid-cycle ovulatory window during the luteal phase. “Within-subject analysis revealed a significant but small pattern of a mid-cycle peak and a luteal decrease at the aggregate level.” The authors note that at the individual level, there was “substantial variability between the direction and magnitude of the testosterone-cycle pattern.” The authors conclude that “salivary testosterone levels show a small trend towards a mid-cycle peak compared to the earlier follicular phase and the later luteal phase of the menstrual cycle when looking at the aggregate across all participants,” but overall, “menstrual patterns of testosterone appear subtle and not systematic across individuals."
Maimoun, et al. (2011) studied the diagnosis process of 5-alpha reductase deficiency. The study included 55 patients with srd5A2 gene mutations. The authors found a “wide spectrum” of phenotypes, including clitoromegaly in 49.1% of participants, microphallus with various degrees of hypospadias in 32.7% of participants, female external genitalia in 7.3% of participants and isolated micropenis in 3.6% of participants. Overall, “over 72% of patients were considered for 5α-reductase deficiency diagnosis when the testosterone/dihydrotestosterone cutoff was ten."
Imperato-McGinley, et al. (1986) studied the clinical criteria used to diagnosis three infants with 5-alpha reductase deficiency. Initially, “basal plasma testosterone to dihydrotestosterone ratios were significantly elevated in two of the three affected infants, and increased markedly in all three infants after administration of hCG.” The authors note that urinary etiocholanolone to androsterone ratios could not be accurately measured in infants, so the diagnosis was confirmed by using gas chromatography/mass spectrometry to measure urinary tetrahydrocortisol (THF) to 5 alpha-tetrahydrocortisol (5 alpha-THF) ratios. “The affected infants had THF/5 alpha-THF ratios comparable to ratios in adult carrier males and significantly lower than ratios in adult homozygotes.” The authors concluded that 5 alpha reductase is detectable at infancy.
Balagamage et al. (2025) conducted a case series of fourteen indiviudals: seven were assigned male gender, six were assigned female gender, and one as non-binary. Most individuals present in the immediate postnatal period, ranging up until 13 years of age. The most common reason for referral was ambiguous genitalia observed at birth. Nine individuals underwent a hCG stimulation test. Six had their hCG stimulation test in infancy, two during the pre-pubertal period (4 and 7 years) and one at the initiation of puberty (12 years). Seven of the nine demonstrated an elevated stimulated T: DHT ratio >10 (median: 15; range: 10.7–66.5); sensitivity 77.78 %. Urinary steroid profiling was performed in 13 affected individuals, including those with T:DHT ratios below 10, and all had decreased ratios of 5α:5β reduced urinary steroid metabolites, predominantly tetra-hydrocortisol and tetra-hydro-corticosterone, suggestive of 5-alpha reductase deficiency. Genetic analysis confirmed SRD5A2 deficiency in all patients. The most commonly reported SRD5A2 gene variants were p.(Arg246Gln), p.(Glu200Lys), and p.(Arg227). No significant correlations were observed between stimulated T:DHT ratios and age at presentation or external masculinisation score. The hCG stimulation test appears less sensitive than urinary steroid profiling in establishing the diagnosis of SRD5A2 deficiency.
Guidelines and Recommendations
The Endocrine Society (ES)-Androgen Deficiency Testosterone Therapy
The ES, in updated 2018 guidelines concerning testosterone therapy and hypogonadism in males, summarized their recommendations with respect to testosterone testing with the following:
- Recommendation 1.1: “We recommend diagnosing hypogonadism in men with symptoms and signs of testosterone deficiency and unequivocally and consistently low serum total testosterone and/or free testosterone concentrations (when indicated).” (Level 1+++)
- Recommendation 1.2: “We recommend against routine screening of men in the general population for hypogonadism.” (Level 1++)
- Recommendation 1.3: “In men who have hypogonadism, we recommend distinguishing between primary (testicular) and secondary (pituitary–hypothalamic) hypogonadism by measuring serum luteinizing hormone and follicle-stimulating hormone concentrations.” (Level 1+++)
- Recommendation 1.4: “In men with hypogonadism, we suggest further evaluation to identify the etiology of hypothalamic, pituitary, and/or testicular dysfunction.” (Level 2++)
- Recommendation 3.1: “In hypogonadal men who have started testosterone therapy, we recommend evaluating the patient after treatment initiation to assess whether the patient has responded to treatment, is suffering any adverse effects, and is complying with the treatment regimen.” (Ungraded Good Practice Statement)
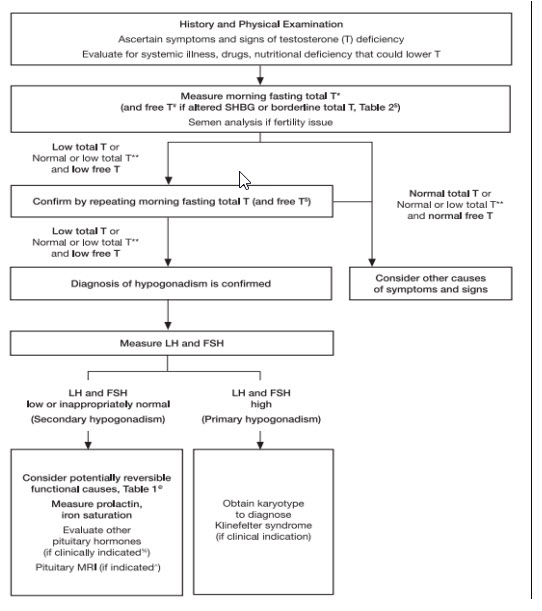
Within the explanations and technical comments of the recommendations, the ES specifically states, “Clinicians should not use direct analog-based free testosterone immunoassays, as they are inaccurate.” Moreover, recommendations state that serum total testosterone testing is preferred and should be performed on two separate days after fasting since testosterone concentrations can vary due to many circumstances. For men who initially test low TT, who test near the lower limit, or who have conditions associated with altering SHBG, then free testosterone can be measured either by using an accepted algorithm based on the TT, SHBG, and albumin concentrations or by direct equilibrium dialysis methods rather than the use of immunoassays. As for bioavailable testosterone testing, the ES states, “Measuring bioavailable [testosterone] concentrations using ammonium sulfate precipitation is technically challenging. Furthermore, there are no detailed studies (similar to those described previously that relate FT [free testosterone] concentrations to manifestations of [testosterone] deficiency) that use bioavailable [testosterone] concentrations.” Men beginning hormone replacement therapy should have their serum testosterone and hematocrit levels measured initially to establish a baseline and then, depending on the therapy, have the levels measured again three to six months later. While on testosterone therapy, the TT and hematocrit levels should be checked annually thereafter. Concerning secondary hypogonadism, serum prolactin and either serum ferritin or iron saturation measurements are recommended to check for the possibility of reversibility of the condition. The testing algorithm also recommends testing serum LH and FSH to differentiate primary and secondary hypogonadism. The algorithm for testing for hypogonadism is shown in the figure below:
Hypothalamic Amenorrhea: An Endocrine Society Clinical Practice Guideline
Testosterone testing in addition to other endocrine laboratory tests is recommended as part of an initial endocrine assessment for women with clinical hyperandrogenism in the evaluation of suspected functional hypothalamic amenorrhea (FHA). FHA is a condition of anovulation, in which the ovary fails to release an egg during the menstrual cycle and has been correlated with stress, weight loss, and excessive exercise.
Polycystic Ovary Syndrome (PCOS)
Relative to the diagnosis of PCOS, the ES identifies three criteria that may be evaluated: androgen excess, ovulatory dysfunction, and polycystic ovaries. Two of the three criteria are sufficient for diagnosis, and if both clinical criteria are met, they do not recommend testing for androgen excess. Androgen excess is characterized by elevated serum androgen levels such as elevated total, bioavailable, or free serum testosterone levels. Considering that serum testosterone levels are variable and there is poor standardization of assays, the Task Force recommends familiarity with local assays and does not define an absolute level that is diagnostic of PCOS or other causes of hyperandrogenism.
Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons
The ES published guidelines suggesting that testosterone level monitoring is suggested at baseline and every 6-12 months during suppression of puberty treatment protocol in gender-dysphoric/gender-incongruent persons. The laboratory monitoring of testosterone levels is also suggested at baseline and every 6-12 months during induction of puberty protocol. Measurement of serum testosterone levels is suggested every three months until levels are in the normal physiologic male range during the monitoring of transgender males on gender-affirming hormone therapy. Testosterone testing is also needed midway between injections for monitoring of testosterone enanthate/cypionate injections, alternatively peak and trough levels could be measured to ensure levels remain in the normal male range. For parenteral testosterone undecanoate, testosterone should be measured just before the following injection. For transdermal testosterone, the testosterone level can be measured no sooner than after one week of daily application (at least 2h after application). For monitoring transgender females on gender-affirming hormone therapy, measurement of serum testosterone is indicated every three months (Hembree et al., 2017).
The American College of Obstetricians and Gynecologists
In 2018, the ACOG released guidelines on the clinical management of polycystic ovary syndrome (PCOS). In its suggested evaluation of patients with PCOS, the ACOG recommends having a physical, laboratory testing, and an ultrasound examination to confirm the polycystic ovaries. With regards to hormone testing, it includes “documentation of biochemical hyperandrogenemia” by “total testosterone and sex hormone binding globulin or bioavailable and free testosterone,” but notes to conduct testing that would exclude other causes of hyperandrogenism, such as thyroid dysfunction and hyperprolactinemia. ACOG includes TSH, prolactin, and 17-hydroxyprogesterone as hormones to measure to exclude other causes. The ACOG (2018) also acknowledges that “there is no standardized testosterone assay in the United States and the sensitivity and reliability in the female ranges are often poor.”
Regarding Müllerian Agenesis, ACOG writes that the initial evaluation of a patient without a uterus “may include the following laboratory tests: testosterone level, FSH level, and karyotype."
In 2019, ACOG released a guideline regarding the “screening and management of the hyperandrogenic adolescent.” In it, they state that the diagnosis of hyperandrogenism can be based on clinical symptoms or measurement of serum androgens. However, they recommend against monitoring serum androgens. This guideline was reaffirmed in 2024.
The ACOG recommends identifying clinical symptoms of androgen excess during the initial evaluation. In the proposed algorithm for evaluation, ACOG recommends two separate batteries of hormone tests depending on type of menses. For regular menses, ACOG lists free and TT, DHEAS (dehydroepianandrosterone sulphate), and 17OHP (17-α-hydroxyprogesterone) as hormones that may be tested. For irregular menses, ACOG lists prolactin, LH, FSH, TSH, and the three previously mentioned hormones. ACOG also notes that PCOS may be one of the diagnoses if both androgen excess and irregular menses are identified. This guideline was reaffirmed in 2024.
American Urological Association (AUA)
The AUA published guidelines concerning the evaluation and management of testosterone deficiency in 2018 and was reaffirmed in 2024. Five recommendations are given concerning the diagnosis of testosterone deficiency:
- “Clinicians should use a total testosterone level below 300 ng/dL as a reasonable cut-off in support of the diagnosis of low testosterone. (Moderate Recommendation; Evidence Level: Grade B)
- The diagnosis of low testosterone should be made only after two total testosterone measurements are taken on separate occasions with both conducted in an early morning fashion. (Strong Recommendation; Evidence Level: Grade A)
- The clinical diagnosis of testosterone deficiency is only made when patients have low total testosterone levels combined with symptoms and/or signs. (Moderate Recommendation; Evidence Level: Grade B)
- Clinicians should consider measuring total testosterone in patients with a history of unexplained anemia, bone density loss, diabetes, exposure to chemotherapy, exposure to testicular radiation, HIV/AIDS, chronic narcotic use, male infertility, pituitary dysfunction, and chronic corticosteroid use even in the absence of symptoms or signs associated with testosterone deficiency. (Moderate Recommendation; Evidence Level: Grade B)
- The use of validated questionnaires is not currently recommended to either define which patients are candidates for testosterone therapy or to monitor symptom response in patients on testosterone therapy. (Conditional Recommendation; Evidence Level: Grade C)”
Other recommendations by the AUA concerning adjunctive testing in males include the following:
- In patients with low testosterone, clinicians should measure serum luteinizing hormone levels (Strong Recommendation; Evidence Level: Grade A)
- Serum prolactin levels should be measured in patients with low testosterone levels combined with low or low/normal luteinizing hormone levels (Strong Recommendation; Evidence Level: Grade A)
- Patients with persistently high prolactin levels of unknown etiology should undergo evaluation for endocrine disorders (Strong Recommendation; Evidence Level: Grade A)
- Serum estradiol should be measured in testosterone deficient patients who present with breast symptoms or gynecomastia prior to the commencement of testosterone therapy. (Expert Opinion)
- Men with testosterone deficiency who are interested in fertility should have a reproductive health evaluation performed prior to treatment. (Moderate Recommendation; Evidence Level: Grade B)
- Prior to offering testosterone therapy, clinicians should measure hemoglobin and hematocrit and inform patients regarding the increased risk of polycythemia. (Strong Recommendation; Evidence Level: Grade A)
- PSA should be measured in men over 40 years of age prior to commencement of testosterone therapy to exclude a prostate cancer diagnosis. (Clinical Principle)."
American Academy of Pediatrics (AAP) —Choosing Wisely Initiative
As a part of the Five Things Physicians and Patients Should Question series of the Choosing Wisely initiative of the American Board of Internal Medicine (ABIM) foundation, the AAP states the following: “Avoid ordering LH and FSH and either estradiol or testosterone for children with pubic hair and/or body odor but no other signs of puberty.” Further, “premature adrenarche is usually the diagnosis and does not involve activation of the pituitary- gonadal axis but is due to an early increase in adrenal androgens. DHEA-S levels are elevated for age but do not alter the management of this common and generally benign condition."
European Academy of Andrology (EAA)
The EAA published guidelines concerning management of bone health in males and testing in Andrology, a journal jointly published by the EAA and the American Society of Andrology. Recommendations include the following:
- “We recommend having serum total testosterone measured twice on a morning blood sample.” (Level 1+++)
- “We recommend measuring again total testosterone and SHBG if only a single measurement documenting low testosterone is available. LH and prolactin are useful to better characterize hypogonadism.” (Level 1+++)
- “We do not recommend routine measurement of serum estradiol.” (Level 1++)
- “We suggest measuring estradiol only when a validated mass spectrometry-based method is available and in rare cases in which severe estrogen deficiency is suspected.” (Level 2++)
Within the evidence and rationale behind the recommendations, the EAA goes on to state, “We suggest using calculated free testosterone when needed, based on the measurement of total serum testosterone, SHBG, and albumin... It can easily be obtained using online available calculators (see Appendix 2 [of (Rochira, et al. (2018)] for Web links). Commercially available kits for direct measurement of free testosterone should not be used due to their poor accuracy and reliability.” Concerning other hormones, the EEA states, “all patients with documented low serum testosterone consulting with hypogonadal symptoms should receive a biochemical evaluation of their gonadal status, with measurement of serum total testosterone, SHBG, luteinizing hormone (LH), follicle-stimulating hormone (FSH), and prolactin."
The EAA also published clinical practice guidelines regarding gynecomastia evaluation and management. The EAA recommended testing several hormones for gynecomastia including “testosterone (T), estradiol (E2), sex hormone-binding globulin (SHBG), luteinizing hormone (LH), follicular stimulating hormone (FSH), thyroid stimulating hormone (TSH), prolactin, human chorionic gonadotropin (hCG), alpha-fetal protein (AFP), liver and renal function tests."
The EAA recently published clinical practice guidelines on investigation, treatment, and monitoring of functional hypogonadism in males to provide certain recommendations:
- “We recommend against universal screening for hypogonadism in middle-aged or older men, by structured interviews or questionnaires and/or random total T measurements.
- We recommend that the clinical diagnosis of functional hypogonadism should be confirmed by measurement of serum total T with a well validated assay on fasting morning (before 11 am) blood samples obtained on two different days.
- Functional hypogonadism should be diagnosed only after exclusion of organic causes of hypogonadism. In addition, to morning total T, luteinizing hormone (LH) should be measured in all patients with suspected functional hypogonadism to differentiate between the primary and secondary causes.
- We recommend either measuring or calculating free T (fT), in addition to total T, in patients with conditions that alter sex hormone-binding globulin (SHBG) and when total T concentrations are in the borderline range (~8-12 nmol/L) if the clinical suspicion of hypogonadism is strong.”
Canadian Urological Association (CUA) and Canadian Society of Endocrinology and Metabolism (CSAM)
The CUA and CSAM endorsed joint guidelines published in the CMAJ in 2015. The following recommendations were given concerning hormone testing in males for testosterone deficiency syndrome:
- “We recommend a thorough history and physical examination, instead of the exclusive reliance on standard questionnaires, to identify patients requiring biochemical testing (strong recommendation; moderate-quality evidence).”
- “The initial biochemical test should be total testosterone level measured in serum samples taken in the morning; determinations of bioavailable testosterone or free testosterone should be restricted to patients with equivocally low total testosterone levels (strong recommendation; high-quality evidence).”
- “We recommend that sample collection for testosterone measurement occur between seven am and eleven am, or within three hours after waking in the case of shift workers (strong recommendation; moderate-quality evidence).”
- “Testosterone levels should be measured with the use of testosterone assays traceable to internationally recognized standardized reference material; commercial assays should be certified by the testosterone standardization program of the US Centers for Disease Control and Prevention (strong recommendation; high-quality evidence).”
- “Measurement of sex hormone–binding globulin with calculated free or bioavailable testosterone should be restricted to men with symptoms of testosterone deficiency and equivocally low testosterone levels (strong recommendation; moderate-quality evidence).”
- “We recommend investigation for secondary or reversible causes of hypogonadism in all men with testosterone deficiency syndrome (strong recommendation; moderate-quality evidence).”
- “We recommend investigation for testosterone deficiency syndrome and treatment with testosterone in men with anemia or sarcopenia of undetermined origin (strong recommendation; moderate-quality evidence).”
- “We recommend assessment of response and adverse effects at three and six months after onset of therapy (strong recommendation; high-quality evidence).”
- “Testosterone levels should be assessed at three and six months after onset of therapy and then annually thereafter if stable (weak recommendation; low-quality evidence).”
European Association of Urology (EAU)
In the 2014 EAU guidelines concerning the treatment of castration-resistant prostate cancer, the EAU states, “Follow-up after ADT should include analysis of PSA and testosterone levels, and screening for cardiovascular disease and metabolic syndrome."
The EAU released guidelines on sexual and reproductive health and expressed the following recommendations for diagnosis of hypogonadism:
- “Check for concomitant diseases, drugs and substances that can interfere with testosterone production/action.
- Measure total testosterone in the morning (between 07.00 and 11.00 hours) and in the fasting state, with a reliable laboratory assay.
- Repeat total testosterone on at least two separate occasions when < 12 nmol/L and before starting testosterone therapy.
- Use 12 nmol/L total testosterone (3.5 ng/mL) as a reliable threshold to diagnose late onset hypogonadism (LOH).
- Measure sex hormone-binding globulin and free testosterone calculation when indicated.
- Analyse luteinising hormone and follicle-stimulating hormone serum levels to differentiate between the different types of hypogonadism.
- Measure prolactin (PRL) levels if evidence of low sexual desire (or other suggestive signs/ symptoms) and secondary hypogonadism is present.
- Perform pituitary magnetic resonance imaging (MRI) in secondary hypogonadism, with elevated PRL or symptoms specific of a pituitary mass and/or presence of other anterior pituitary hormone deficiency.
- Perform pituitary MRI in secondary severe hypogonadism (total testosterone < 6 nmol/L)..
- Screen for late onset hypogonadism (LOH) only in symptomatic men.
- Do not use structured interviews and self-reported questionnaires for systematic screening for LOH as they have a low specificity.”
The EAU recommends that the standard and most accurate method for testosterone serum testing is liquid chromatography-tandem mass spectrometry (LC-MS/MS). Standardized automated platform immuno-assays are reliable techniques to measure testosterone; however, only LC-MS/MS can provide an accurate measurement of FT (fT) levels. When diagnosing LOH, the EAU recommends measuring fasting and morning (7-11am) TT, noting to “(consider PRL measurement if low desire or other suggestive symptoms are present,” “consider SHBG and free-T calculations when indicated,” consider LH when T deficiency pathophysiology must be investigated.” There is uncertainty as to what threshold of fT level indicates hypogonadism, but some data indicates that fT levels below 225 pmol/L is associated with hypogonadism.
National Comprehensive Cancer Network
Within the algorithm concerning the systemic therapy for castration-naïve disease, the NCCN says to “document castrate level of testosterone if clinically indicated” when assessing progression along with the physical exam and PSA every three to six months. The NCCN also states to “continue ADT [androgen deprivation therapy] to maintain castrate serum levels of testosterone (<50 ng/dL).” Additional recommendations state, “close monitoring of PSA and testosterone levels and possibly imaging is required when using intermittent ADT, especially during off-treatment periods, and patients may need to switch to continuous ADT upon signs of disease progression."
The NCCN also published some guidance regarding assessment of hormones for neuroendocrine and adrenal tumors. For pituitary tumors, they list serum prolactin and LH/FSH; for “suspected or confirmed adrenocortical carcinoma”, they list "screen for hypercortisolemia (± Cushing syndrome) and primary aldosteronism” and “adrenal androgens (DHEAS, androstenedione, testosterone, 17-hydroxyprogesterone)”; for hypercortisolemic Cushing’s Syndrome, they list “screen for hypercortisolemia (± Cushing syndrome) with 1 of the following tests: 1 mg overnight dexamethasone suppression test, 2–3 midnight salivary cortisols, [or] 24-hour urinary free cortisol” and “plasma ACTH [adrenocorticotropic hormone] in AM if confirmed hypercortisolemia (± Cushing syndrome)."
International Late Effects of Childhood Cancer Guideline Harmonization Group (IGHG) & PanCareSurFup (PCSF) Consortium
Within the guidelines and recommendations issued in 2017 by the IGHG and the PCSF Consortium for patients with possible impaired spermatogenesis, it is recommend that “Clinical measurement of testicular volume and of follicle-stimulating hormone and inhibin B might be reasonable for the identification of impaired spermatogenesis in survivors treated with potentially gonadotoxic chemotherapy or radiotherapy potentially exposing the testes in whom semen analysis has been declined or is not possible and who desire assessment about possible future fertility. Be aware of the diagnostic limitations of these tests that may result in false positives or false negatives (level B evidence).” With respect to patients with possible testosterone deficiency, “Measurement of testosterone concentration in an early morning blood sample at clinically appropriate intervals is reasonable in post pubertal survivors treated with radiotherapy potentially exposing the testes to 12 Gy or more or with TBI (expert opinion). In the presence of clinical signs of hypogonadism, or of previous low-normal or borderline testosterone concentrations, or if it is not possible to obtain an early morning blood sample, it is reasonable to measure luteinising hormone concentration in addition to testosterone (expert opinion)” (Skinner et al., 2017).
American Society of Reproductive Medicine (ASRM)
The ASRM, in collaboration with the Society for Male Reproduction and Urology, released a committee opinion on the diagnostic evaluation of sexual dysfunction in the male partner in the setting of infertility. The publication recommends the following for the detection of erectile dysfunction: “A physical examination should include blood pressure and the calculation of body mass index, as well as an assessment for signs of testosterone deficiency. Morning serum testosterone should be assayed, as should glucose and hemoglobin A1c levels, as well as lipid profile measurements, as indicated."
State and Federal Regulations, as applicable
Many labs have developed specific tests that they must validate and perform in house. These laboratory-developed tests (LDTs) are regulated by the Centers for Medicare and Medicaid (CMS) as high-complexity tests under the Clinical Laboratory Improvement Amendments of 1988 (CLIA ’88). LDTs are not approved or cleared by the U. S. Food and Drug Administration; however, FDA clearance or approval is not currently required for clinical use.
Billing/Coding/Physician Documentation Information
This policy may apply to the following codes. Inclusion of a code in this section does not guarantee that it will be reimbursed. For further information on reimbursement guidelines, please see Administrative Policies on the Blue Cross Blue Shield of North Carolina web site at www.bcbsnc.com. They are listed in the Category Search on the Medical Policy search page.
Applicable service codes: 82040, 82642, 82670, 82681, 84270, 84402, 84403, 84410
BCBSNC may request medical records for determination of medical necessity. When medical records are requested, letters of support and/or explanation are often useful, but are not sufficient documentation unless all specific information needed to make a medical necessity determination is included.
Scientific Background and Reference Sources
Gill-Sharma MK. Testosterone Retention Mechanism in Sertoli Cells: A Biochemical Perspective. The open biochemistry journal. 2018;12:103-112. doi:10.2174/1874091X01812010103
Bhasin S, Brito JP, Cunningham GR, et al. Testosterone Therapy in Men With Hypogonadism: An Endocrine Society* Clinical Practice Guideline. The Journal of Clinical Endocrinology & Metabolism. 2018;103(5):1715-1744. doi:10.1210/jc.2018-00229
CDC. Hormones Certified Assays and Participants. Updated November 17, 2025.https://www.cdc.gov/clinical-standardization-programs/php/hormones/list-of-hormone-certified-assays.html
Kinter KJ, Anekar AA. Biochemistry, Dihydrotestosterone. StatPearls Publishing, Treasure Island (FL); 2023.
Schulster M, Bernie AM, Ramasamy R. The role of estradiol in male reproductive function. Asian J Androl. May-Jun 2016;18(3):435-40. doi:10.4103/1008-682x.173932
Hammond GL, Bocchinfuso WP. Sex hormone-binding globulin/androgen-binding protein: steroid-binding and dimerization domains. The Journal of steroid biochemistry and molecular biology. Jun 1995;53(1-6):543-52. doi:10.1016/0960-0760(95)00110-l
Longcope C. Adrenal and gonadal androgen secretion in normal females. Clin Endocrinol Metab. May 1986;15(2):213-28. doi:10.1016/s0300-595x(86)80021-4
Burger HG. Androgen production in women. Fertil Steril. Apr 2002;77 Suppl 4:S3-5. doi:10.1016/s0015-0282(02)02985-0
Teede HJ, Misso ML, Costello MF, et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Human reproduction (Oxford, England). Sep 1 2018;33(9):1602-1618. doi:10.1093/humrep/dey256
Carnegie C. Diagnosis of hypogonadism: clinical assessments and laboratory tests. Rev Urol. 2004;6 Suppl 6(Suppl 6):S3-8.
Nassar GN, Leslie SW. Physiology, Testosterone. StatPearls. StatPearls Publishing LLC.; 2023.
Snyder PJ. Clinical features and diagnosis of male hypogonadism. Wolters Kluwer. Updated September 10, 2025. https://www.uptodate.com/contents/clinical-features-and-diagnosis-of-male-hypogonadism
Meldrum DR, Gambone JC, Morris MA, Esposito K, Giugliano D, Ignarro LJ. Lifestyle and metabolic approaches to maximizing erectile and vascular health. International journal of impotence research. Mar-Apr 2012;24(2):61-8. doi:10.1038/ijir.2011.51
Stanworth RD, Jones TH. Testosterone for the aging male; current evidence and recommended practice. Clinical interventions in aging. 2008;3(1):25-44. doi:10.2147/cia.s190
Hassanabad MF, Fatehi M. Androgen Therapy in Male Patients Suffering from Type 2 Diabetes: A Review of Benefits and Risks. Current diabetes reviews. Jul 31 2018;doi:10.2174/1573399814666180731125724
Mohammed M, Al-Habori M, Abdullateef A, Saif-Ali R. Impact of Metabolic Syndrome Factors on Testosterone and SHBG in Type 2 Diabetes Mellitus and Metabolic Syndrome. Journal of diabetes research. 2018;2018:4926789. doi:10.1155/2018/4926789
Wang A, Arver S, Flanagan J, et al. Dynamics of testosterone levels in patients with newly detected glucose abnormalities and acute myocardial infarction. Diabetes & vascular disease research. Oct 3 2018:1479164118802543. doi:10.1177/1479164118802543
Corona G, Rastrelli G, Di Pasquale G, Sforza A, Mannucci E, Maggi M. Endogenous Testosterone Levels and Cardiovascular Risk: Meta-Analysis of Observational Studies. The journal of sexual medicine. Sep 2018;15(9):1260-1271. doi:10.1016/j.jsxm.2018.06.012
Molina-Vega M, Munoz-Garach A, Damas-Fuentes M, Fernandez-Garcia JC, Tinahones FJ. Secondary male hypogonadism: A prevalent but overlooked comorbidity of obesity. Asian J Androl. Jul 3 2018;doi:10.4103/aja.aja_44_18
Viana A, Jr., Daflon AC, Couto A, Neves D, de Araujo-Melo MH, Capasso R. Nocturnal Hypoxemia is Associated With Low Testosterone Levels in Overweight Males and Older Men With Normal Weight. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine. Dec 15 2017;13(12):1395-1401. doi:10.5664/jcsm.6832
Goldman AL, Bhasin S, Wu FCW, Krishna M, Matsumoto AM, Jasuja R. A Reappraisal of Testosterone's Binding in Circulation: Physiological and Clinical Implications. Endocrine reviews. Aug 1 2017;38(4):302-324. doi:10.1210/er.2017-00025
CDC. Steroid Hormones Standardization Programs. Updated May 14, 2024. https://www.cdc.gov/clinical-standardization-programs/php/hormones/index.html
Mezzullo M, Fazzini A, Gambineri A, et al. Parallel diurnal fluctuation of testosterone, androstenedione, dehydroepiandrosterone and 17OHprogesterone as assessed in serum and saliva: validation of a novel liquid chromatography-tandem mass spectrometry method for salivary steroid profiling. Clinical chemistry and laboratory medicine. Aug 28 2017;55(9):1315-1323. doi:10.1515/cclm-2016-0805
Andersson CR, Bergquist J, Theodorsson E, Strom JO. Comparisons between commercial salivary testosterone enzyme-linked immunosorbent assay kits. Scandinavian journal of clinical and laboratory investigation. Dec 2017;77(8):582-586. doi:10.1080/00365513.2017.1339231
Welker KM, Lassetter B, Brandes CM, Prasad S, Koop DR, Mehta PH. A comparison of salivary testosterone measurement using immunoassays and tandem mass spectrometry. Psychoneuroendocrinology. Sep 2016;71:180-8. doi:10.1016/j.psyneuen.2016.05.022
van der Veen A, van Faassen M, de Jong WHA, van Beek AP, Dijck-Brouwer DAJ, Kema IP. Development and validation of a LC-MS/MS method for the establishment of reference intervals and biological variation for five plasma steroid hormones. Clin Biochem. Jun 2019;68:15-23. doi:10.1016/j.clinbiochem.2019.03.013
Star-Weinstock M, Dey S. Development of a CDC-certified total testosterone assay for adult and pediatric samples using LC–MS/MS. Clinical Mass Spectrometry. 2019;13:27-35.
Sun G, Xue J, Li L, et al. Quantitative determination of human serum testosterone via isotope dilution ultra performance liquid chromatography tandem mass spectrometry. Mol Med Rep. Aug 2020;22(2):1576-1582. doi:10.3892/mmr.2020.11235
Lynch KL. CLSI C62-A: A New Standard for Clinical Mass Spectrometry. Clinical Chemistry. 2016;62(1):24-29. doi:10.1373/clinchem.2015.238626
Yun Y-M, Botelho JC, Chandler DW, et al. Performance Criteria for Testosterone Measurements Based on Biological Variation in Adult Males: Recommendations from the Partnership for the Accurate Testing of Hormones. Clinical Chemistry. 2012;58(12):1703-1710. doi:10.1373/clinchem.2012.186569
Vesper H, Botelho J, Wang Y. Challenges and improvements in testosterone and estradiol testing. Invited Review. Asian Journal of Andrology. March 1, 2014 2014;16(2):178-184. doi:10.4103/1008-682x.122338
Khashchenko E, Uvarova E, Vysokikh M, et al. The Relevant Hormonal Levels and Diagnostic Features of Polycystic Ovary Syndrome in Adolescents. J Clin Med. Jun 11 2020;9(6)doi:10.3390/jcm9061831
Dalmiglio C, Bombardieri A, Mattii E, et al. Analytical performance of free testosterone calculated by direct immunoluminometric method compared with the Vermeulen equation: results from a clinical series. Hormones (Athens). Jun 2024;23(2):313-319. doi:10.1007/s42000-023-00522-x
Vermeulen A, Verdonck L, Kaufman JM. A critical evaluation of simple methods for the estimation of free testosterone in serum. The Journal of clinical endocrinology and metabolism. Oct 1999;84(10):3666-72. doi:10.1210/jcem.84.10.6079
Sartorius G, Ly LP, Sikaris K, McLachlan R, Handelsman DJ. Predictive accuracy and sources of variability in calculated free testosterone estimates. Annals of clinical biochemistry. Mar 2009;46(Pt 2):137-43. doi:10.1258/acb.2008.008171
Zakharov MN, Bhasin S, Travison TG, et al. A multi-step, dynamic allosteric model of testosterone's binding to sex hormone binding globulin. Molecular and cellular endocrinology. Jan 5 2015;399:190-200. doi:10.1016/j.mce.2014.09.001
Travison TG, Vesper HW, Orwoll E, et al. Harmonized Reference Ranges for Circulating Testosterone Levels in Men of Four Cohort Studies in the United States and Europe. The Journal of clinical endocrinology and metabolism. Apr 1 2017;102(4):1161-1173. doi:10.1210/jc.2016-2935
Shukla A, Sharda B, Bhardwaj S, et al. Association Between Serum Testosterone and Serum PSA Among Men With and Without Partial Androgen Deficiency. Indian Journal of Clinical Biochemistry. 2018:1-5.
Sachdev S, Cucchiara AJ, Snyder PJ. Prostate Specific Antigen Concentrations in Response to Testosterone Treatment of Severely Hypogonadal Men. Journal of the Endocrine Society. 2020;doi:10.1210/jendso/bvaa141
Zitzmann M, Nieschlag E, Traish A, Kliesch S. Testosterone Treatment in Men with Classical vs. Functional Hypogonadism: A 9-Year Registry. Journal of the Endocrine Society. 2019;3(1)doi:10.1210/js.2019-SUN-222
Cauley JA, Ellenberg SS, Schwartz AV, Ensrud KE, Keaveny TM, Snyder PJ. Effect of testosterone treatment on the trabecular bone score in older men with low serum testosterone. Osteoporos Int. Nov 2021;32(11):2371-2375. doi:10.1007/s00198-021-06022-1
Stern J, Casto K. Salivary testosterone across the menstrual cycle. Horm Behav. Aug 2024;164:105608. doi:10.1016/j.yhbeh.2024.105608
Maimoun L, Philibert P, Cammas B, et al. Phenotypical, biological, and molecular heterogeneity of 5α-reductase deficiency: an extensive international experience of 55 patients. The Journal of clinical endocrinology and metabolism. Feb 2011;96(2):296-307. doi:10.1210/jc.2010-1024
Imperato-McGinley J, Gautier T, Pichardo M, Shackleton C. The diagnosis of 5 alpha-reductase deficiency in infancy. The Journal of clinical endocrinology and metabolism. Dec 1986;63(6):1313-8. doi:10.1210/jcem-63-6-1313
Balagamage C, Igbokwe R, Cole T, et al. The diagnostic value of stimulated androgen ratios in 5-alpha reductase type 2 (SRD5A2) deficiency: a case series and review of the literature. Journal of Pediatric Endocrinology and Metabolism. 2025;38(9):922-930. doi:doi:10.1515/jpem-2025-0195
Gordon CM, Ackerman KE, Berga SL, et al. Functional Hypothalamic Amenorrhea: An Endocrine Society Clinical Practice Guideline. The Journal of clinical endocrinology and metabolism. May 1 2017;102(5):1413-1439. doi:10.1210/jc.2017-00131
Legro RS, Arslanian SA, Ehrmann DA, et al. Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline. The Journal of clinical endocrinology and metabolism. Dec 2013;98(12):4565-92. doi:10.1210/jc.2013-2350
ACOG. ACOG Practice Bulletin No. 194: Polycystic Ovary Syndrome. Obstet Gynecol. Jun 2018;131(6):e157-e171. doi:10.1097/aog.0000000000002656
ACOG. ACOG Committee Opinion No. 728: Müllerian Agenesis: Diagnosis, Management, And Treatment. Obstet Gynecol. Jan 2018;131(1):e35-e42. doi:10.1097/aog.0000000000002458
ACOG. Screening and Management of the Hyperandrogenic Adolescent: ACOG Committee Opinion, Number 789. Obstet Gynecol. Oct 2019;134(4):e106-e114. doi:10.1097/aog.0000000000003475
Mulhall JP, Trost LW, Brannigan RE, et al. Evaluation and Management of Testosterone Deficiency. American Urological Association. https://www.auanet.org/guidelines-and-quality/guidelines/testosterone-deficiency-guideline
AAFP. Avoid ordering luteinizing hormone- and follicle-stimulating hormone and either estradiol or testosterone for children with pubic hair and/or body odor but no other signs of puberty. https://www.aafp.org/pubs/afp/collections/choosing-wisely/352.html
Rochira V, Antonio L, Vanderschueren D. EAA clinical guideline on management of bone health in the andrological outpatient clinic. Andrology. Mar 2018;6(2):272-285. doi:10.1111/andr.12470
Kanakis GA, Nordkap L, Bang AK, et al. EAA clinical practice guidelines-gynecomastia evaluation and management. Andrology. Nov 2019;7(6):778-793. doi:10.1111/andr.12636
Corona G, Goulis DG, Huhtaniemi I, et al. European Academy of Andrology (EAA) guidelines on investigation, treatment and monitoring of functional hypogonadism in males. Andrology. 2020;8(5):970-987. doi:10.1111/andr.12770
Morales A, Bebb RA, Manjoo P, et al. Diagnosis and management of testosterone deficiency syndrome in men: clinical practice guideline. Cmaj. Dec 8 2015;187(18):1369-1377. doi:10.1503/cmaj.150033
Heidenreich A, Bastian PJ, Bellmunt J, et al. EAU guidelines on prostate cancer. Part II: Treatment of advanced, relapsing, and castration-resistant prostate cancer. European urology. Feb 2014;65(2):467-79. doi:10.1016/j.eururo.2013.11.002
Salonia, Bettocchi, Carvalho, et al. European Association of Urology: Sexual and Reproductive Health. 2025;
NCCN. Prostate Cancer, Version 2.2026. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
NCCN. Neuroendocrine and Adrenal Tumors Version 2. 2025. https://www.nccn.org/professionals/physician_gls/pdf/neuroendocrine.pdf
Skinner R, Mulder RL, Kremer LC, et al. Recommendations for gonadotoxicity surveillance in male childhood, adolescent, and young adult cancer survivors: a report from the International Late Effects of Childhood Cancer Guideline Harmonization Group in collaboration with the PanCareSurFup Consortium. The Lancet Oncology. Feb 2017;18(2):e75-e90. doi:10.1016/s1470-2045(17)30026-8
ASRM. Diagnostic evaluation of sexual dysfunction in the male partner in the setting of infertility: a committee opinion. Fertil Steril. Nov 2023;120(5):967-972. doi:10.1016/j.fertnstert.2023.07.001
Avalon review 12/2018
Specialty Matched Consultant Advisory Panel 6/2020
Specialty Matched Consultant Advisory Panel 6/2021
Medical Director review 6/2021
Medical Director review 1/2022
Medical Director review 12/2022
Medical Director review 1/2024
Medical Director review 12/2024
Medical Director review 1/2026
Policy Implementation/Update Information
For Policy Titled: Hormonal Testing in Adult Males
1/1/2019 New policy developed. BCBSNC will provide coverage for Testosterone Testing when it is determined to be medically necessary because the medical criteria and guidelines are met. Medical Director review. Policy noticed on 1/1/2019 for effective date 4/1/2019. (mco)
4/16/19 Policy renamed from Testosterone to Hormonal Testing in Males. For information regarding androgen deficiency in females, see policy titled “Hormonal Testing in Females AHS G2161.” Policy Description and Guidelines updated. The following changes were made to clinical criteria: Added “after fasting” to statement concerning the measurement of total serum testosterone in according to 2018 Endocrine Society and 2018 EAA guidelines. For the measurement of free testosterone, the wording was changed to state: “For males with hypogonadism, gynecomastia, and/or other forms of testicular hypofunction, measurement of serum free testosterone using a medically accepted algorithm based on total serum testosterone, sex hormone-binding globulin (SHBG), and/or albumin is considered medically necessary if total testosterone is confirmed as borderline or low.” Added statement that free testosterone and/or bioavailable testosterone as source of initial testosterone testing (i.e. total testosterone should be determined first based on 2018 Endocrine Society guidelines). Clarified “total” testosterone testing for gender dysphoric/gender incongruent individuals. Added statement that measurement of serum LH, FSH, and prolactin ONCE to differentiate primary and secondary male criteria (based on multiple guidelines). Added statement for males with gynecomastia, serum estradiol testing ONCE prior to initiating testosterone therapy. (2018 AUA guidelines). Added statement that serum LH, FSH, prolactin, and/or estradiol in males in other situations. Added statement that serum dihydrotestosterone (DHT) testing in males is investigational (new 2019 CPT code) except in cases for testing of 5-alpha reductase deficiency. Added CPT codes 82040, 82642, 82670, 83001, 83002, 84146, and 84270 to Billing/Coding section. References updated. (an)
9/10/19 Policy coverage criteria updated. Removed once in a lifetime limit from #10 in the when covered section. Added to when covered section #11; stating measurement of serum prolactin, LH, FSH, growth hormone (GH), thyroid stimulating hormone (TSH), and adrenocorticotropic hormone (ACTH) are medically necessary in the diagnosis and management of pituitary adenoma and in the diagnosis of hypopituitarism. Added to #5 of when not covered section, “with ambiguous genitalia, hypospadias, or microphallus.” Coding table removed. Codes listed and added 84443, 83003, and 82024. (eel)
10/1/19 Medical Director review 8/2019. Reviewed by Avalon 2nd Quarter 2019 CAB. (eel)
10/29/19 Wording in the Policy, When Covered, and/or Not Covered section(s) changed from Medical Necessity to Reimbursement language, where needed. (gm)
2/11/20 Reviewed by Avalon 4th Quarter CAB. No changes to policy. (eel)
7/14/20 References updated. Specialty Matched Consultant Advisory Panel review 6/17/2020. No changes to policy. (eel)
2/9/21 Reviewed by Avalon 4th Quarter CAB, Medical Director Review 1/2021. “This policy only addresses coverage of hormonal testing in adult males (age 18 years and older)” added to Description section. When covered criteria updated for clarity by removing language referencing non-adult males from item 1 and 4. References to Notes updated with addition of Note 1 to coverage section. Added code 82681. Policy name changed from Hormonal Testing in Males AHS-G2013 to Hormonal Testing in Adult Males AHS-G2013. Description, Policy Guidelines, and References updated. (bb)
7/1/21 Specialty Matched Consultant Advisory Panel review 6/16/2021. Medical Director review. No change to policy statement. (lpr)
Policy Re-titled: Testosterone Testing
2/8/22 Policy re-titled, “Testosterone Testing” for consistency with Avalon. Reviewed by Avalon 4th Quarter 2021 CAB, Medical Director Review 1/2022. Removed CPT codes: 82021, 83001, 83001, 83003, 84146, 84443. When covered criteria updated: removed language related to codes removed; Added Reimbursement is allowed for testing for serum total testosterone* (See Note 1) symptomatic females being evaluated for conditions associated with androgen excess (e.g., polycystic ovary syndrome and functional hypothalamic amenorrhea). The technology used for testing should be sensitive enough to detect the low concentrations normally found in females.” When not covered criteria updated: Changed “males” to “individuals” in item 4; Added Reimbursement is not allowed for testing for serum testosterone for the identification of androgen deficiency in women. Policy Guidelines and References updated. Description updated, changed “males” to “individuals”. (tt)
6/30/22 Added the following, “For Policy Titled: Hormonal Testing in Adult Males” to implementation section for clarity. (tt)
Policy Re-titled: Testosterone
2/7/23 Policy re-titled, “Testosterone” for consistency with Avalon. Reviewed by Avalon 4th Quarter 2022 CAB. Policy statement updated to reflect name change. Description, Policy Guidelines, and References updated. Coverage criteria, Note 1 and Note 2 updated for clarity. Added Note 3 and Note 4. Medical Director Review 12/2022 (tt)
2/21/24 Reviewed by Avalon 4th Quarter 2023 CAB. Minor grammar changes to Policy Guidelines. References updated. Medical Director review 1/2024. No changes to coverage criteria. (rp)
2/26/25 Reviewed by Avalon 4th Quarter 2024 CAB. Policy Guidelines and References updated. No changes to coverage criteria. Medical Director review 12/2024. (tt)
5/6/26 Reviewed by Avalon 1st Quarter 2026 CAB. Policy Guidelines and References updated. No changes to coverage criteria. Medical Director review 1/2026. (tt)
Blue Cross and Blue Shield of North Carolina does not discriminate on the basis of race, color, national origin, sex, age or disability in its health programs and activities. Learn more about our non-discrimination policy and no-cost services available to you.
Information in other languages: Español 中文 Tiếng Việt 한국어 Français العَرَبِيَّة Hmoob ру́сский Tagalog ગુજરાતી ភាសាខ្មែរ Deutsch हिन्दी ລາວ 日本語
© 2026 Blue Cross and Blue Shield of North Carolina. ®, SM Marks of the Blue Cross and Blue Shield Association, an association of independent Blue Cross and Blue Shield plans. All other marks and names are property of their respective owners. Blue Cross and Blue Shield of North Carolina is an independent licensee of the Blue Cross and Blue Shield Association.